Hi there. I was recently notified by CVS Caremark that they no longer are covering Vemlidy after we fought hard with the petition in 2019 to put it back on their formulary list. Anyone having this issue and any information on what is being done ? What are y’all doctors next steps in insuring that you remain on the medication vs switching to alternatives ? Thoughts?
Hi I am in the same situation. I would like to know what is being done. I’ve reached out to the Hep B foundation and asked for any suggestions on what I can do. It seems like I can’t do anything.
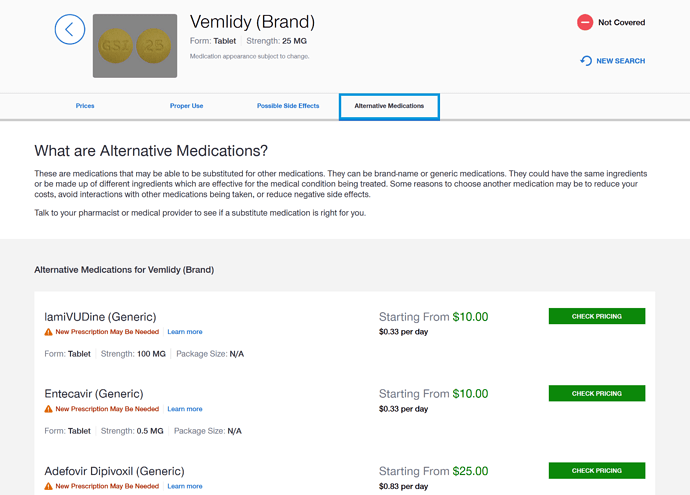
I’m glad this thread came up. I’m with United Healthcare, and will be discussing starting treatment with my new Gastroenterologist, as it was suggested to me by my old one before I moved. Vemlidy is not covered under my health insurance as well. I guess the real question we should be asking is how much better is Vemlidy than the generic alternative, if at all? For United Healthcare, these are the alternatives they suggest to Vemlidy:
Are they any worse than Vemlidy? Is there any harm to changing from Vemlidy, to a generic alternative?
AFAIK lamivudine or adefovir will eventually lead to viral resistance - so in that sense, they are much worse.
Hi @Adam1, @Tulipe72001, and @dhoang1212,
Thanks for bringing up this important issue. I’m not from the US, so perhaps others can comment more on the situation (@Suwang88, @chari.cohen, etc.).
Regarding the switching, the generics that you’ve mentioned below are different types of drug. As mentioned by @mantana, Lamivudine is not recommended due to not being effective and resistance. The 1st line treatments for HBV infection are entecavir (ETV) and tenofovir disaproxil fumarate (TDF, which is most similar to TAF/Vemlidy); both of these are available as generics. You should talk to your health care provider about what is best for your medical background.
Thomas
Thanks for the advice @ThomasTu
This is a very important topic for patients impacted, I am one of them. My doctor submitted an appeal for me, fortunately the insurance company approved so I can stay on Vemlidy for one year. History: I was on Entecavir for 7 years but eventually developed resistance to this drug. For patients just starting new treatment, Entecavir is a good option.
Hi can I ask what insurance you have? The only reason I am asking is because my doctor is also doing an appeal and wondering if they will reject it. My insurance is with CVS Caremark. Jackie
My Insurance company is Aetna, and the formulary manager is also CVS Caremark. My doctor submitted the appeal on Monday and he called me and told me that the insurance approved today. Best of luck!
Oh wow! That was quick . I had spoken with my doctor who said she was going to submit an appeal. That was last week and I haven’t heard back . I also have Aetna with CVS Caremark. Did you do anything on your end to get a quick approval ? Any tips for the rest of us? I’ve tried TDF but had bad side effects so they switched me to Vemlidy . I’m so upset as there was a successful petition in 2019 to have CBD Caremark add Vemlidy back into its formulary . Thought that was the end of it until now . Can’t believe they took it off again.
Hi, I am not exactly sure what the doctor wrote in the appeal letter, but he did mention to me before that there were unsuccessful appeal cases, so it is not a guarantee that an appeal will be successful for everyone. I talked to his office staff about my case, I guess these helps: 1. I developed resistance to Entecavir so I can’t go back to it. 2. Viread has more adverse effects on bone density/renal system, since I enter menopause and am more susceptible to bone loss, Viread is not a good option for me either.
I did some research on where I can buy cheaper Vemlidy and found out that Gilead is only selling it for approximately USD52 per 30 days usage in China. India pharmacies sells cheaper version of Vemlidy too. I was planning to explore that option if my appeal was denied ( there are quite some reputable China on line pharmacies which conducts remote prescription). I am still very upset that Vemlidy is being taken away from the US patients whose country invented the drug. We should continue to pressure the insurance companies/Giread to resolve this issue.
That is definitely fast. I would also like to know what your doctor wrote that got approved so fast. Thank you
Hi- Thanks so much for posting this. If possible, could you send an email to Catherine.Freeland@hepb.org ? My colleagues and I can try to help with this issue! Please reach out!
Hi! Thank you for replying to this post. I will reach out to the email you provided . It’s unbelievable what is happening after all the publicity in 2019 to get it back on the formulary . I’m wondering what the justifications that all these big pharmaceutical providers have in denying coverage . Is this a common thing in your experience with brand name medications with no generics available?
Do you have the online link to Chinese online pharmacies that sells Vemlidy? I need to explore that option too. Thanks in advance.
Hi Thomas! If to switch to TDF, what is the equivalent mg to Vemlidy 25mg? Is there any related science article? Thanks!
Hi @SeanL,
@availlant has a post about this: Possible side-effects from antiviral therapy - #95 by availlant
300mg TDF is the equivalent amount (though this can sometimes be reported as 245mg depending on how that company is calculating the mass of the active compound). Your pharmacist will know as it is quite a standard dose.
Thomas
Not a medical expert here but from my general understanding, resistance with Entecavir is rare. Did you skip doses or have them at irregular timing?
@ThomasTu
I switched from Viread to Vemlidy a few years ago. If I switch back to Viread now, should I worry about drug resistance issue with Viread?
Hi @Tahlah,
In both, the active compound is tenofovir, so there should not be an issue with drug resistance if the antivirals are still working.
Thomas