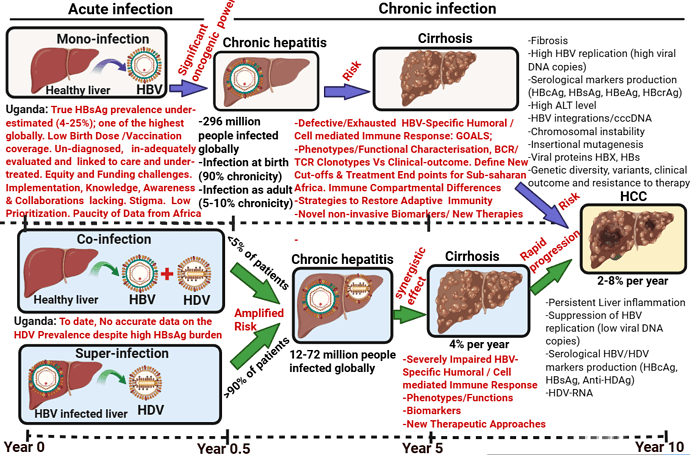
Chronic HBV and HDV Simultaneity: frequent, complex and deadly
Hepatitis delta infection (hepatitis Delta virus (HDV) disease may present a significant health burden among viral liver diseases particularly in low-income countries, but accurate epidemiological information remains unknown due to lack of epidemiological studies. In recent meta-analysis, an estimated 12 million people worldwide have been infected with HDV but the large gaps in diagnosis in especially high-prevalence areas and populations might underestimate this number being backed by a meta-analysis indicating 50–72 million Hepatitis B virus (HBV) carriers may be coinfected with HDV. The exact prevalence and estimated number of HDV patients is still a subject of debate for several reasons, including unreliable assessment of infection and a lack of real-world screening. HDV is a satellite virus that can infect and multiply only in the presence of HBV surface antigens (HBsAg) and its the least prioritised human virus, even though coinfection (when HBV and HDV are present at the same time) and superinfection (when both viruses are present in sequence) may cause serious health problems. HDV significantly enhances the risk of hepatocellular carcinoma (HCC) and is the key determinant in the rapid progression of cirrhosis in HBsAg-positive people compared to HBV alone. HDV infection in chronic HBV patients is a severe form of hepatitis that often ends in liver cancer, cirrhosis, and death. Estimates place HDV as the cause of 20% of HBV-related HCC and 18% of liver cirrhosis. Lack of access to healthcare in low- and middle-income countries often results in underdiagnosis of the disease, preventing early detection and effective treatment.
Double reflex testing of HDV in all HBsAg+ is crucial in low-income countries:
Despite 40 years of epidemiological surveys the number of infected people with HDV remains undefined 48-60millio, 62-72million, 15-20million 12million etc. These widely different figures emphasise the heterogeneity of the HDV report and lack of sufficient quality data. Possible reasons for this include inadequate screening, technical limitations, the limited awareness among healthcare providers, the requirement for costly equipment and specialized training, and a lack of access to reliable tests in regions with poor medical infrastructure, lack of data to confirm ongoing virus replication on the epidemiology and clinical data on HDV. HDV as satellite of HBV causes severe liver disease. In certain low-income nations, the hepatitis Delta virus and HBV co-occurrence could pose a serious medical burden, for example in Uganda where the prevalence of HBsAg is estimated to be greater than 4.3% among adults and could be as high as 25% in some regions of Uganda which is one of the highest prevalence globally, but the true estimate is still inaccurate. The highest HBsAg-positivity rates are observed in West Nile, Northern and Northeastern sub regions of Uganda which are chronic HBV-endemic and highly impoverished regions of Uganda. Especially in rural places, non-existent or irregular diagnostic examinations are the main restriction and reason for underestimation. Major barriers to HBV elimination goal in Uganda are under-funding both domestically and internationally, low prioritization and collaborations compared to HIV, TB and malaria, low birth dose/vaccination coverage in newborns and adolescents, lack of knowledge, awareness, testing and linkage to care, high stigmatisation and discrimination levels for those found infected, under-evaluation and treatments of cases with often test kits, antivirals and birth doses running out of stock. Despite this disproportionately high HBV burden in Uganda, the accurate prevalence of HDV infection in HBsAg+ fraction is missing. Thus, lack of universal testing of HBsAg-positive individuals for anti-HD is certainly the single most important reason for underestimating the prevalence of HDV infections. We propose that integration of routine HBV/HDV screening in existing maternal HIV /Syphilis routine care as well in general population would be an important strategy for epidemiological studies, clinical diagnostics, biomarker discovery, evaluating new agents on a backbone of Interferon/ Nucleos(t)ide analogues (NUC) therapy especially in regions that currently lack access to reliable HBV and HDV testing. Novel diagnostic tool/biomarkers that can guide treatment cessation and monitor intrahepatic viral clearance, that is non-invasive, standardized, amenable to automation, robust and reproducible are needed. New biomarkers would be useful to better characterize patients mono and co-infected patients who do not fit into defined phases of chronic HBV; predict response to IFN at an early stage, so that those with high probability of response are prioritized; identify patients who could discontinue NUCs without risk of relapse; assess target engagement and antiviral activity in patients receiving novel molecular entities (NME) and to predict HBsAg response, i.e. functional cure, in patients receiving novel antiviral or immunomodulatory agents. How HBV mono and HBV/HDV co-occurrence modulates antiviral immune response would be highly relevant to design new therapeutic approaches. Also, significant genetic heterogeneity of HBV genotype A in sub-Saharan Africa, underscores the relevance of genetic analysis of HBV strains from this part of the continent and this is crucial for understanding their impact on disease outcome, immune response, response to therapy and routes of HBV spread globally as well to define New cut-offs and Treatment End points in context dependent manner.
B cells have largely been neglected and understudied
Previous research has understandably focused on restoring the function of exhausted virus-specific Tcells in chronic HBV infections but there has been a relative lack of appreciation for the role played by the Bcell pool and virus-specific Memory Bcell clones in controlling HBV infection. Indeed, several humoral immune studies points towards their key role: An in-vivo data shows that anti-HBsAg antibodies (HBsAb) can recognize circulating HBsAg and clear infectious HBV particles; the presence of HBsAb in serum is considered an indicator of the resolution of CHB; moreover HBsAg-seroconversion is regarded as a successful treatment endpoint; HBeAg-seroconversion heralds the transition between clinical phases and comes with improved immune control; B-cell depleting treatments, such as rituximab (targeting CD20 and CD52), may lead to potential fatal HBV flares, even in patients with a resolved infection suggesting essential role of antibodies to HBV in maintain HBV under immune surveillance; An unexpected HBsAg clearance frequently encountered after bone marrow transplantation from vaccinated donors to CHB recipient. These data suggest that B-cell-based humoral immunity may for long-term sustain HBV control. Emerging evidence has highlighted the defective function of HBsAg-specific B cells in CHB patients, which might contribute to the incapability to achieve a functional cure of CHB. The underlying mechanism responsible for impaired B cell function in Chronic HBV mono and HDV-co-infected patients remains elusive. The role of immune compartments in shaping the HBV-specific response is a critical piece of the puzzle in both acute and chronic infection in exploring therapeutic angles targeting these compartments to achieve functional cure.
Recent HBV Research activity in Uganda
STUDY SUMMARY
| Study Title | Prevention of mother-to-child transmission of hepatitis B virus by establishing an antenatal screening and viral load testing unit and tenofovir disoproxil fumarate treatment in Lira, Northern Uganda |
|---|---|
| Study Design | Pilot observational study |
| Study Participants and setting | We aimed to build capacity for antenatal hepatitis B surface antigen (HBsAg) screening in pregnant women at the gynaecology departments of Lira University Hospital and Lira Regional Referral Hospital, followed by viral load testing in HBsAg-positive participants via q‑PCR at Lira University. The proposed screening approach to identify HBV-infected pregnant mothers in the region and facilitate their referral to care centers. We screened a total of 800 pregnant women, and for those with an HBV DNA viral load ≥10,000 IU/mL were provide antiviral treatment with tenofovir disoproxil fumarate to prevent mother-to-child transmission of HBV. |
| Intervention(s) | Hepatitis B virus mono-infected women with a viral load ≥10,000 IU/mL during weeks 24 to 28 of gestation received tenofovir disoproxil fumarate treatment up to 12 weeks after delivery. |
| Primary objective | We assessed the implementation of the HBV DNA viral load threshold of ≥10,000 IU/mL for initiating TDF treatment in pregnant women during gestation week 24 or beyond in order to prevent HBV mother-to-child transmission in Lira, Northern Uganda. |
| Secondary objectives | * We determined the prevalence and risk factors associated with hepatitis B virus infection among pregnant women attending the antenatal clinic in Lira University Hospital and Lira Regional Referral Hospital. |
- We successfully optimised linkage to care and engagement in the recommended care after receiving a positive HBsAg test during the antenatal period.
- We assessed and enhanced knowledge and perceptions of hepatitis B, with the aim of reducing stigma and improving awareness among pregnant women.
- We assessed the viral load and effectiveness of the TDF therapy.|
|Primary Endpoint|We determined the proportion of hepatitis B virus mono-infected women who meet the criteria for TDF treatment using a viral load cut-off of ≥10,000 IU/mL, as well as the proportion of TDF treated pregnant women achieving a viral load of ≤10,000 IU/mL at the time of childbirth.|
|Secondary Endpoint(s)|* We determined the proportion of HBsAg-positive pregnant women in two antenatal clinics in Lira, and its associated risk factors. - We determined the proportion of pregnant women who are successfully linked to care after receiving a positive HBsAg test during the antenatal period.
- We assessed the perceptions and knowledge of pregnant women towards HBV infection.
- We determined the proportion of HBsAg-positive pregnant women with a viral load ≥10,000 IU/mL who adhere to TDF treatment.|
|Planned Sample Size|We screened 800 pregnant women for hepatitis B in the antenatal clinics of Lira University Hospital and Lira Regional Referral Hospital.|
|Follow up duration|HBsAg-positive pregnant women with a viral load of ≥10,000 IU/mL were followed-up from weeks 24 to 28 of gestation until week 12 after delivery.|
Due to limited funding and other funding difficulties, we couldn’t continue with the study/intervention in Northen Uganda. Data analysis, curation and manuscript is underway.
Am open to opinions, inputs, suggestions, comments, strategies and future research collaborations from community, experts and Funders on the discussed matter etc
Kind regards
Tom Adomati (Ugandan)
Postdoctoral Research Scientist- Viral Hepatitis (Immunology)
University of Antwerp